Why magnet only attract iron not others?
Why magnet only attract iron not others?
Actually Magnets attract all objects, but Something that is strongly attracted to a magnet is said to have a high permeability. Iron and steel are two examples of materials with very high permeability, and they are strongly attracted to magnets. Liquid oxygen is an example of something with a low permeability, and it is only weakly attracted to a magnetic field. Water has such a low permeability that it is actually repelled by magnetic fields. Everything has a measurable permeability: people, gases and even the vacuum of outer space.
Permeability
In electromagnetism, permeability is the degree of magnetization of a material that responds linearly to an applied magnetic field.
Hence, If you succed to make a very big magnet you will see that it has a impact on other things also
Example- CERN
http://www.interactions.org/cms/?pid=102…
http://www.local6.com/news/4783604/detai…
http://news.nationalgeographic.com/news/…
So, Actually when you want to see the proof that magnets attract other things also see the earth itself the Gravitational force is the earth’s magnet magnetic force and it attracts unlimited things.
Hope it Helps!!!
Source(s):
http://www.physicsforums.com/archive/ind…
http://en.wikipedia.org/wiki/Magnet
http://en.wikipedia.org/wiki/Permeabilit…
when a magnet attracts iron the atoms inside the iron aling themselves in straight chains where as in other substance
the atomms are alinged in closed chains. If you break these chains then atom will aling themselves in straight chains and then they would be attracted towards the magnets but for this you require huge magnetic force.
see there are 5 elements in nature knwn as ferromagnetic..these are the elements which are magnetic and can be conversely attracted by magents…iron is included in this list of ferromagnetic elements..so it is attracted…
hope u gt d basic thing
Have you ever heard of a ferromagnetic metal? That is a type of metal that responds strongly to magnetic fields. There are only a few of them – some are common metals, like iron, cobalt, and nickel. Others are very rare, and have funny names like gadolinium, Terbium, Dysprosium, and Holmium. Anyway, almost all magnets are made out of these metals or mixes (alloys) of them. By the way, a nickel (the coin) is made out of 3/4 copper and only 1/4 nickel – this mixture is not very magnetic at all, but a pure piece of nickel would be.
So, why are ferromagnetic metals attracted by magnetic fields? Like all other material things, these metals are made up of atoms. But the atoms are very special, because they have what is called a magnetic
dipole. Atoms have “positive” charges at the center, called protons. They also have negative charges whizzing around in circles around the positive charges – these negative ones are called “electrons.” When
electrons go in a circle (really really fast, by the way!) they make a magnetic field. The important thing to understand is this: most kinds of atoms have a bunch of electrons going around in different ways
and they make a bunch of little magnetic fields that point in different
directions and cancel each other out. Metals like iron are different –
they have electrons arranged so that the magnetic fields do NOT cancel
out. Instead, each little iron atom has a magnetic field.
There is ANOTHER important thing about iron. A piece of iron has billions of billions of atoms, so many you could never count them all,
even if you were as old as the earth! If all of these atoms had their
little magnetic field pointed in different directions, they still would not amount to anything. But with a piece of iron, the atoms arrange themselves into little groups called “domains.” Each iron
atom in a domain has its magnetic dipole field aligned perfectly with all the other atoms in that domain. They just form that way when the iron is made.
Now, if you bring an ordinary piece of iron close to another magnet, the magnetic field will try to spin these “domains” around so they are aligned with it. Imagine this: you and all your classmates are outside at recess, all running around and facing different directions. Then the teacher yells at you and tells you to listen. Everyone turns around and faces the teacher. That’s sort of what these magnetic domains
do.
Anyway, if some of the domains turn around, the iron becomes magnetic. The more they align, the more magnetic the iron gets. If you get it magnetic enough, it stays that way EVEN when you take it away from the
first magnet. That explains why you can use one magnet to make another one out of an ordinary piece of iron. (You can’t do this forever, though, cause the new one you make is a lot weaker than the first one.)
You might think it is strange that you can actually get these domains to move – but they really move! People have done experiments where they hook up a REALLLY sensitive microphone to the iron – and you can HEAR
the domains turning. (Sounds like popcorn).
2. These electrons are what makes an atom magnetic. How many electrons
there are, and how they are moving determines whether the magnetic
field adds up to something big or not.
3. Iron atoms are special, because they are magnetic (have magnetic
dipoles) and their magnetic fields (their dipoles) line up perfectly
with all of their neighbors in the same domain
4. When a piece of iron comes near a magnetic field (like another magnet)
, these domains actually turn around around to try to align with that
field. This gives the piece of iron a big magnetic field, too, and then
the two magnets attract.
5. If enough domains turn around and align, the iron will still stay
a magnet after the first magnet goes away.
So now you might wonder about all the rules for the electrons, and
how you can explain how they move and such. That is a complicated
question that requires a lot of math to answer, but the answer is VERY INTERESTING! !
 How to link Mobile Number with Aadhaar using OTP
How to link Mobile Number with Aadhaar using OTP  Difference between Aadhaar and Social Security Number (US)
Difference between Aadhaar and Social Security Number (US)  Do you Know GK Today Series,Interesting Facts and figures
Do you Know GK Today Series,Interesting Facts and figures 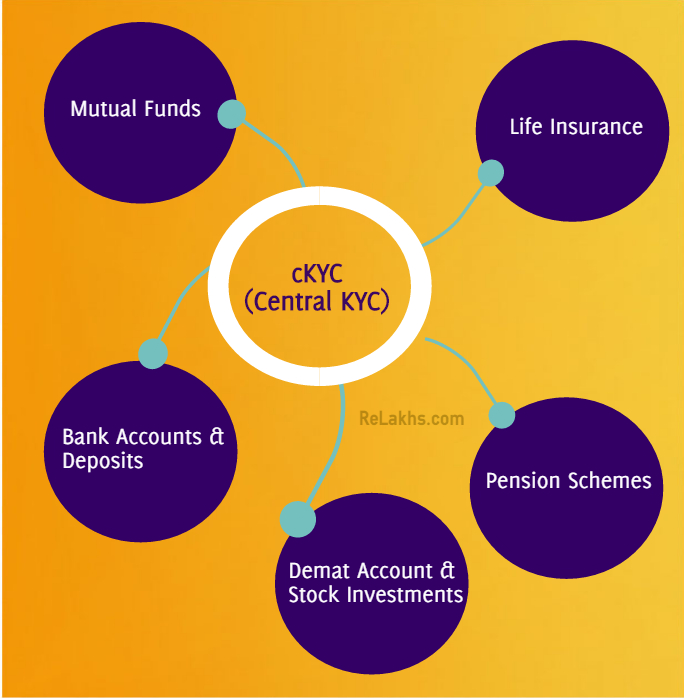 The Central KYC Registry or Know your Customer Registry
The Central KYC Registry or Know your Customer Registry  Statue of Unity and Top Tallest Statues in the World
Statue of Unity and Top Tallest Statues in the World  New Fund Offering by SBI MF – SBI Dividend Yield Fund
New Fund Offering by SBI MF – SBI Dividend Yield Fund  Bharti Airtel’s 5G user-base expands
Bharti Airtel’s 5G user-base expands  GK TODAY -HISTORY AT A GLANCE
GK TODAY -HISTORY AT A GLANCE
good explanation